Adapted from a Phlexglobal Innovation Tour webinar, “Crossing the TMF Compliance Gap with Inspection-Ready Access for Closed Studies,” held April 21, 2021. You can view the webinar – available on-demand – to discover how companies are successfully bridging that gap to ensure ongoing inspection-readiness. To learn more about Phlexglobal’s solution for crossing the TMF Compliance Gap, visit PhlexTMF for Viewing, or contact us today.
What is the TMF Compliance Gap?
In a nutshell, the TMF Compliance Gap occurs when your CRO uses their eTMF system to manage your study’s electronic Trial Master File. When the study is live, the eTMF is generally secure and inspectable in the CRO’s eTMF system - especially if they are using an advanced eTMF purpose-built for TMF management.
Once the study is closed, the CRO returns the eTMF to you for the reporting, assessment, and submission phase. Most eTMF systems are not designed or priced for longer-term inspectable storage, and CROs don’t want to be in the business of maintaining closed eTMFs. Your CRO will usually return the final eTMF in portable electronic formats such as DVDs or USB drives, cloud-based file folders, or even printed out on paper.
The likelihood of an inspection remains high during this period, however, and can last several years (see image).

This is the TMF Compliance Gap: Your electronic Trial Master File no longer resides in an easily viewable and inspectable system, during a lengthy timeframe where an inspection is still highly likely. And you still need a secure and accessible place to store the final eTMF that is compliant with all necessary GCP requirements for archiving of study documents.
What’s Wrong With Having the eTMF in Digital Storage Formats Like USB Drives?
When talking about inspectable storage for the Trial Master File after the study is closed, it’s important to clarify the distinction between the eTMF (electronic Trial Master File) and the system that enables and supports that TMF in electronic form. Think of your eTMF system (Phlexglobal’s PhlexTMF software, for example) as the interface into your Trial Master File.
When we talk about an eTMF system, we’re referring to a solution designed solely to easily navigate and manage the Trial Master File while the study is underway, based on the TMF Reference Model standard. The result is improved TMF management and compliance, as well as more streamlined inspections.
Once you transfer the electronic TMF to any storage format, however, those capabilities disappear. In addition, portable electronic media are often lost or misplaced, while cloud-based file folders pose their own set of security and navigation problems. And paper, of course, is practically unworkable in today’s environment of remote inspections.
In our recent webinar on this topic, 58% of respondents said they were receiving the eTMF from their CRO in portable electronic formats such as USB drives or DVDs. Following are some of the often overlooked challenges with these formats:
- Do you have an established and compliant SOP for the eTMF after the study is closed? Who is responsible for it during this period?
- How would you run an inspection with the content in its current format? Can your eTMF still effectively tell the story of the trial for inspectors? How do you keep related study documents together with the final eTMF?
- Is there a process is for reconstructing a navigable eTMF from the files? What is the original navigation scheme?
- Where is the storage media kept? Who has access to it? How is it kept secure, with a continuous chain of custody? How do you prevent files from being changed or overwritten?
- How do you ensure access and readability as technology changes? What about degradation of data over time? Do you have secure and controlled backups?
Note: Cloud-based storage, while answering some of the above problems, is a partial solution at best. It also creates additional challenges such as validation and online access control.
To see why the file and folder structure doesn’t work, consider a typical scenario where an inspector requests all IRB submissions. Having these files organized by site is a fairly common approach. You can imagine the inspector’s reaction when you hand them some USB drives or give them access to your cloud storage. They see the folders illustrated here, with the files nested within the folders (see image).
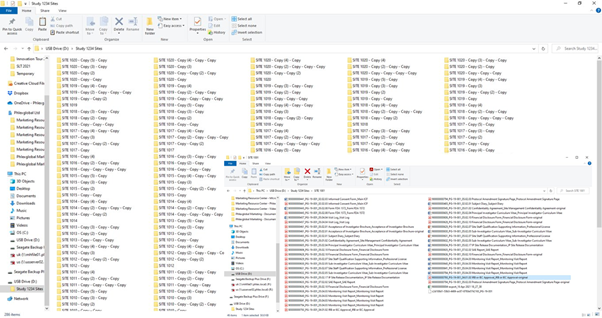
Their reaction? They will either have your team provide the answers they need, requiring a massive amount of additional time and effort, or simply delay the inspection until a better solution is in place.
And even if you have the resources to spare to do the work, doing so only creates additional problems. How do you present the example of all IRB submissions? You have few options other than to go into each folder, locate the correct documents, then copy and paste them somewhere else. You’re taking the data out of the system and creating duplicate files – so which is the verified original? How do you keep track of everything?
The Solution to the TMF Compliance Gap Is In the Metadata
Most eTMF systems are designed around the notion of metadata or descriptive data. Documents are consistently classified according to the TMF Reference Model standard. So in the example above, in a purpose-built eTMF system, it takes only a few minutes to find all the IRB submissions based off the artifact type. What’s more, inspectors are increasingly accustomed to having access to these systems, which further frees up your internal resources.
At Phlexglobal, we found that the primary challenge in solving the TMF Compliance Gap was how to adapt technology designed to manage the eTMF during a live study to a long-term storage solution. This is essentially a new category of eTMF system, retaining the core functionality needed for easily viewing and inspecting the eTMF, yet with an affordable and predictable pricing model designed for longer-term and compliant storage.
A secondary – yet still significant – challenge lay in taking the eTMF files provided by a CRO, regardless of format, and ensuring they are replicated with 100% accuracy in the inspectable eTMF storage system. Here, we were able to draw on the on the extensive expertise of Phlexglobal’s in-house TMF migration team. As a result, we can take the Trial Master File documents from a CRO in any format ranging from paper and portable media to a competitor’s eTMF system, with the associated metadata and audit trail, and put it in our validated system for inspectable storage access.
This turnkey solution, PhlexTMF for Viewing, encompasses software and expert services for inspectable and affordable eTMF storage – turning the industry-wide TMF Compliance Gap into a bridge that any organization can easily cross.




