Introduction
You came back, great! I hope you enjoyed our first blog in this series which was all about the basics of a clinical trial.
In this blog we are going to get into the Trial Master File, which we will just be referring to as the TMF from now on. What is the TMF? Why is it important? And why should anyone care about it?
So lets not stand about, lets get right into it.
TMF, you mean a few trial documents?
The TMF serves a very simple and key role in a clinical trial, it is there to prove and document that the trial was run in a way that met all necessary regulatory requirements and conformed with Good Clinical Practices (GCP).
Now I know what you are thinking, “Ok, sounds easy enough, so why is there and entire industry around generating, maintaining and advancing the TMF”. Well, that is because although it sounds easy when written down like this lets look at what goes into the TMF.
ICH E6 Part 8 describes a list of Essential Documents and was originally brought in back in the 90’s, these include things like:
- Protocol – THE document that describes what the trial is all about and lays out exactly how it will be performed
- Signed Protocol – Signed copies or at least signature pages of the Protocol that show each and every site has read and understood the protocol and what is being asked of them
- Protocol Amendments – I mean you didn’t think that things wouldn’t change at some point did you, these trials can go for years. Amendments are used to keep track of the changes to the trial.
- Regulatory Authorization and Approval – It goes without saying but you can’t just turn up in a company and start handing out new drugs, you need to request to perform the trial in that country and get approval to do so
- Investigator CVs – You need to make sure the people running your trial at the site are capable of such an important role so it makes sense you would do the same thing as when you bring onboard any staff or resource
So above are a few example, and when I say a few I mean a tiny amount compared to the full list.
Lets do a little thought experiment right now. You have a trial so you are going to need a Protocol, that’s one (1) document, you then want to run you trial in 5 countries so you need to produce the authorization document and then receive the approval, that’s 10 documents. Each country has 10 sites so that’s 50 sites in total, 50 Protocol signature pages and 50 Investigator CVs. So that’s what, 111 documents, all coming in at different times and from different places. Then something happens, remember 2020, remember when the whole world had to change. Something like that is going to trigger an amendment, perhaps putting the trial on hold or changes to the way site and patient interaction will happen. So now you need a Protocol Amendment, get it submitted and approved, send out to all your sites, get them to review it and then provide a signature page. We are well over 200 documents and all we have done is send out some documents to say what we will do, we haven’t even done anything yet.
This, right here, is why the TMF is such a critical part of the trial and why it is so complicated.
A TMF needs to be kept for every single clinical trial performed and also needs to be kept well after the study finishes. As you can imagine trying to keep the TMF up to date in a timely manner, making sure quality does not suffer and that it has everything it needs to be completed is a real challenge, and one we will touch in in another blog, don’t want to cause too much excitement in one post now.
What I would like to touch on though is the Reference Model and why it is such a big deal.
The TMF Reference Model, where Too Much Fun meets the TMF
Once upon a time, long, long ago, when the Black Eyed Peas had a feeling, Lady Gaga was coming along and telling us to just dance and we all went to Pandora to run around with blue people with long braids there was something special going on in the world of TMF.
The TMF has been essential for year, but the issue was every single company and every single group did it differently. The essential documents list was helpful, but it wasn’t a complete list, and when it came to how to organize these documents (and this was primarily paper documents stored in files and folders) every one did it in their own way. This caused countless issues, regulators would have to work out how it was being organized and understand what people had decided should be in and what shouldn’t, people moving from companies had to re-learn everything with each new employer wasting potentially years of experience and for the companies themselves trying to maintain their own thing was a costly and time consuming piece of work.
A group of particularly smart folks within this space figured something needed to happen and so along with other like minded individuals set to work on doing something about it. What they came up with was the TMF Reference Model, a standardized set of expected documents in a structure that could be understood along with metadata that just made sense.
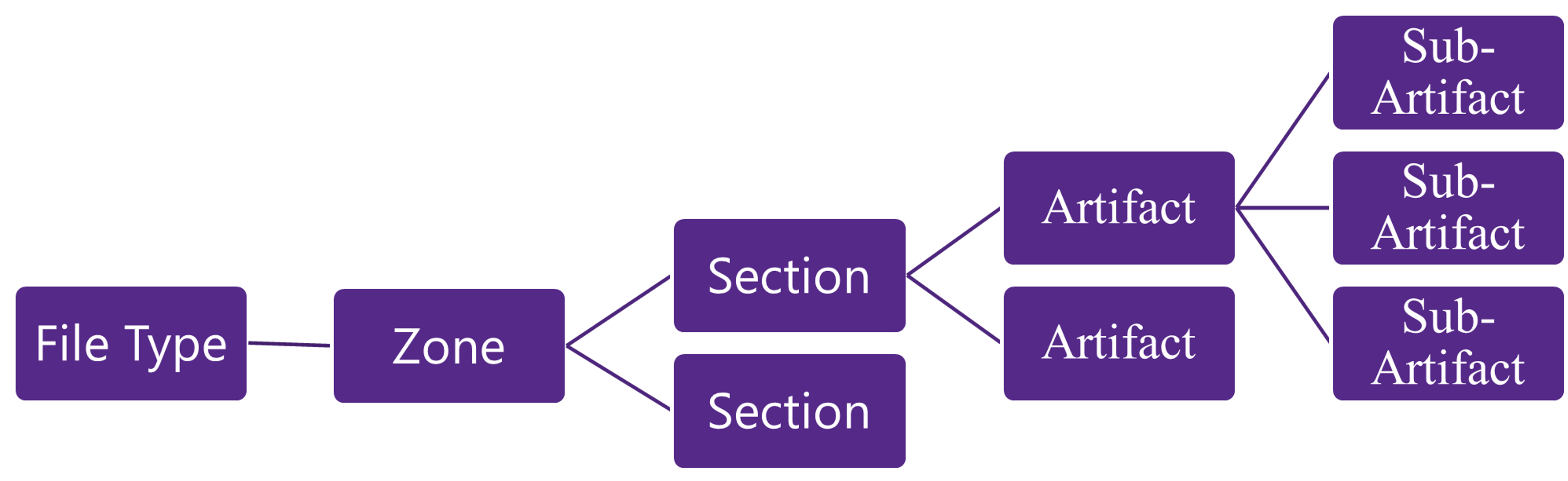
Structure:
A simple hierarchy was created to help people navigate through the TMF and get to the documents they need as simply as possible.
- File Type – This could be Study, Country or Site. Where does the document belong, is it a Study level document like the Protocol, Country level like that Regulatory Approval or Site like the CVs.
- Zone – The highest level of the structure which group all similar types, things like all documents that could be classified as Trial Management, or Site Management or Safety etc. There are 11 Zones.
- Section – A second level of hierarchy to help define in more detail the document
- Artifact – This is the first level that documents or data could be located, it would be a collection of documents such as all the Amendments for that trial.
- Sub Artifact – for those wanting more granularity Sub-Artifacts are a great option, the artifact contains a collection of data whereas the Sub-Artifact can take you further. Rather than just storing all Meeting Materials at the Artifact, Sub-Artifacts can be used to split out into Agendas, Minutes, presentations etc.

The other thing to know is that this structure is re-used over an over, what I mean is that each site has their own structure with all their own documents and data following the hierarchy, as does every Country. You could have hundreds of the same Artifact throughout the study at different File Types and across all your sites included.
Expected Documents and Data:
The Essential Documents list is great but it is still only a small number of the documents you could need. The TMF Reference Model introduced a number of other documents that are more than likely going to be collected and filed in order to maintain the ultimate goal, which is to prove and document that the trial was run in a way that met all necessary regulatory requirements and conformed with GCP. It also helped companies to remove some of the guess work to what could be needed and allow them to be a lot more efficient.
Metadata:
As the world changed and the time of software and digital solutions became more relevant the TMF Reference Model changed and adapted by bringing in suggested metadata that should be assigned to the files stored.
The TMF Reference Model has been amended, adapted and improved over the last 10+ years and has had contributions from well over 1,500 individuals at this point. It is not owned by a single person or company and has always been aimed for use by anyone regardless of if they are a Sponsor or CRO, small start up or Top 10 Pharma company, or still using paper or maybe moved over to a system (they don’t care which one).
I would really advise anyone wanting to know more about the reference model to check out the webpage especially as there are some exciting changes happening now they have become part of the Clinical Data Interchange Standards Consortium (CDISC)
Closing Remarks
It is safe to say that we could write hundreds of blogs on the TMF and its importance to the clinical trial but for now we will stick with this one. I hope it gave you some clarity about what it is, an idea of why it is such a big deal and also why the people that work in it are awesome and incredibly smart in order to manage and maintain all this information.
Have questions about Clinical Trials and the TMF? Watch our "Clinical Trials and TMF 101 Learning Series" webinar here. Or, contact me today if you would like to talk more about anything Clinical Trial and TMF related.
We are now two blogs down in our Clinical Trials and the TMF 101 series, next we are going to talk about Paper vs Electronic and how its more than just storing a PDF on your desktop.
Until then, take care
Rob Jones


